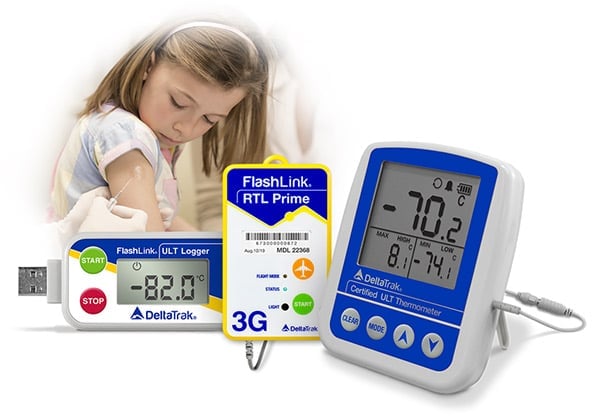
Clinical Trial Solutions
Failure to adequately monitor temperatures during shipping and storage can lead to clinical trials being significantly delayed.
Compromised shipments
Optimum conditions need to be maintained for drugs designated for clinical trials across the supply chain. Ineffective temperature monitoring can impact the whole process.

Human error
Efficient temperature monitoring is crucial before clinical trials begin, but too often human error can lead to mistakes in record keeping or even failure to activate data loggers.

Continuous monitoring
DeltaTrak’s solutions deliver cost-effective, continual monitoring throughout the cold chain and storage stages, delivering accurate records even if the logger is not started.
Selected DeltaTrak Clinical Trial Solutions
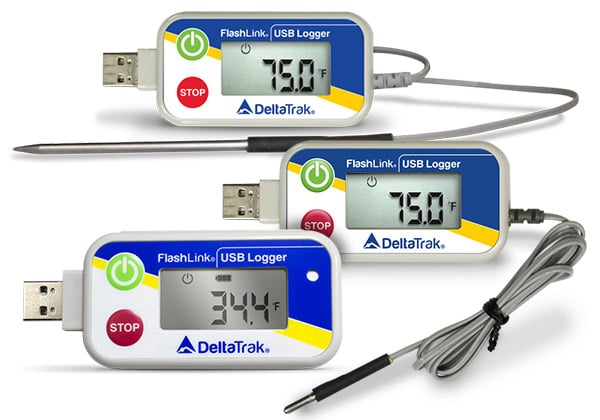
Reusable BLE Data Loggers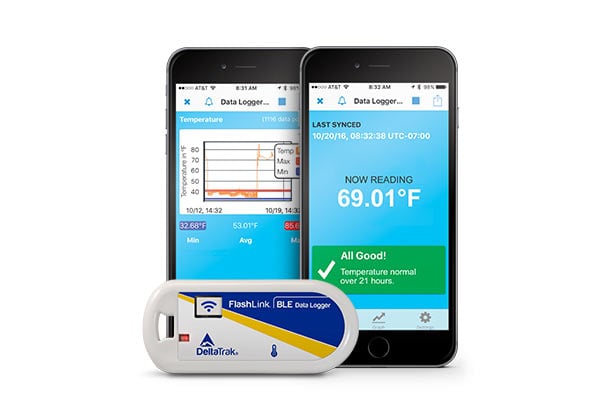 In-Transit PDF Loggers
In-Transit PDF Loggers Reusable Data Loggers
Reusable Data Loggers